Blog
FDA Approves Enhertu + Pertuzumab as First-Line Treatment for HER2-Positive Metastatic Breast Cancer (December 2025)
Samar Elkassas
Feb 12, 2026

Big News for People Living with HER2-Positive Metastatic Breast Cancer:
On December 15, 2025, the U.S. Food and Drug Administration (FDA) approved a new first-line treatment (the first treatment given) for adults with HER2-positive metastatic breast cancer.
The new approved combination is:
Enhertu (fam-trastuzumab deruxtecan)
Plus pertuzumab (brand name: Perjeta)
This is an important step forward and may change how treatment starts for many patients.
Learn more about breast cancer and available treatment options in our full resource guide.
What Does “HER2-Positive” Mean?
Some breast cancers make too much of a protein called HER2. This protein helps cancer cells grow faster.
When a cancer is “HER2-positive,” doctors can use targeted treatments that specifically attack cells with HER2. These treatments have already improved survival significantly over the past two decades.
Now, this new approval brings another powerful option even earlier in treatment.
What Is Enhertu?
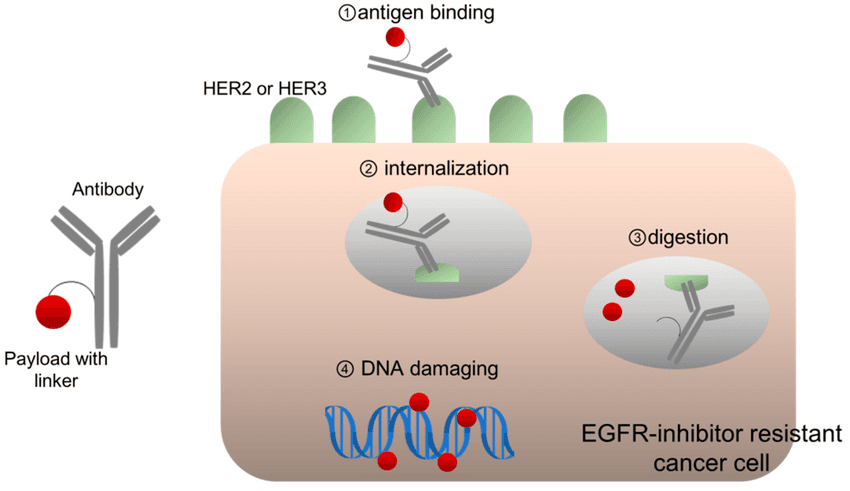
Enhertu is sometimes described as “smart chemotherapy.”
It belongs to a group of medicines called antibody-drug conjugates (ADCs). That means:
One part of the drug acts like a guided missile — it finds HER2-positive cancer cells.
The other part carries chemotherapy directly inside those cells.
This helps deliver treatment more precisely to cancer cells.
Pertuzumab (Perjeta) is another HER2-targeted medicine that works in a different way. Together, they form a powerful combination.
Why Is Starting Treatment So Important?
The first treatment for metastatic breast cancer can shape the entire journey.
Starting with a highly effective option may:
Help control cancer longer
Shrink tumors more deeply
Delay the need for other treatments
Potentially improve quality of life
That’s why this approval is considered a major update.
What Study Led to This Approval?
The FDA approval is based on results from a large clinical trial called:
DESTINY-Breast09
This study looked at using Enhertu-based treatment in people who had not yet received therapy for metastatic HER2-positive breast cancer. The results showed strong benefit, leading to approval.
Are There Side Effects?
Like all cancer treatments, Enhertu + pertuzumab can cause side effects.
Doctors will discuss possible risks with you, including:
Nausea
Fatigue
Low blood counts
Lung inflammation (a rare but serious side effect called interstitial lung disease)
Your oncology team will monitor you closely and help manage any side effects.
Questions You Might Want to Ask Your Doctor
If you or a loved one has HER2-positive metastatic breast cancer, you might ask:
“Is Enhertu + pertuzumab an option for me?”
“How does this compare to the previous standard treatment?”
“What side effects should I expect?”
“How will we monitor my response?”
Every person’s cancer is different. Your treatment plan will be personalized based on your health, prior treatments, and preferences.
The Bottom Line
A new first-line treatment option is now available for HER2-positive metastatic breast cancer.
The approval of Enhertu + pertuzumab represents another step forward in targeted therapy, offering patients a new way to start treatment with precision and power.
If you’re navigating this diagnosis, talk openly with your oncology team about whether this new option may be right for you.
Related Resources
ctDNA Trials in Breast Cancer: How TREAT ctDNA, DARE and Apollo Are Changing Early Intervention
Blog





